Their nucleus is capable of decaying and breaking down into smaller particles. 
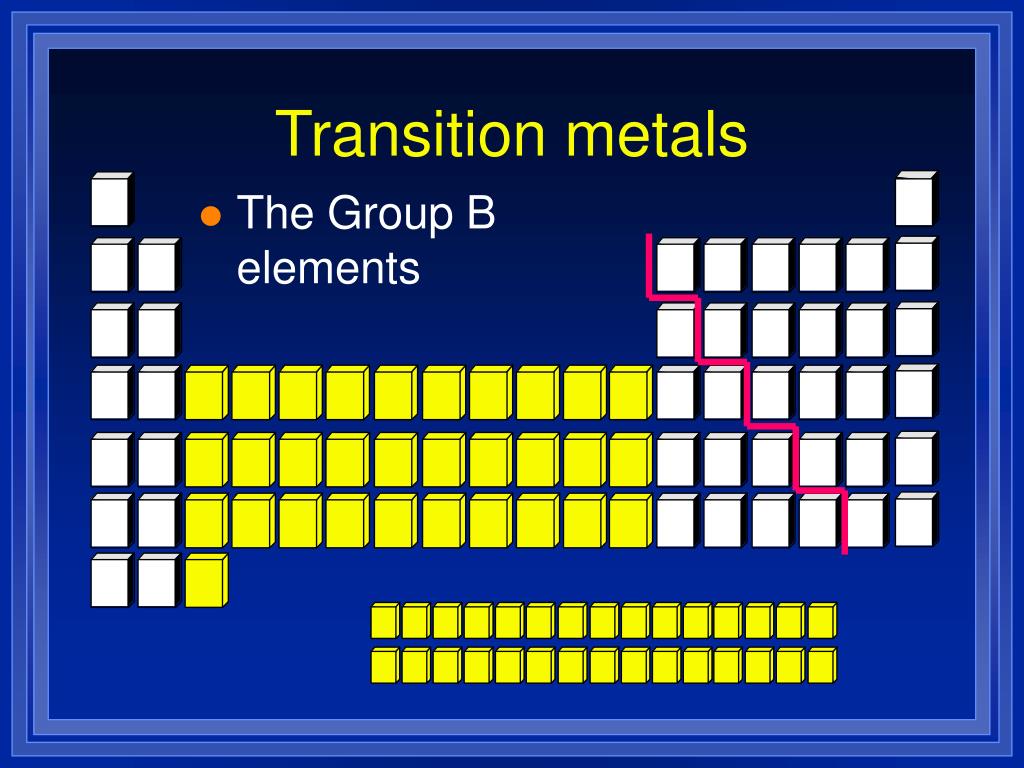
These three main transition series are included in the set of 30 elements often called the d-block transition metals. 222K subscribers Subscribe Subscribed 1.3K Share 290K views 10 years ago MCAT The Periodic Table The definition of a transition metal, and how to write the electron configuration. The actinides definition describes a group of chemical elements that are characterized by their radioactive nature. The third series extends from lanthanum (symbol La, atomic number 57) to mercury (symbol Hg, atomic number 80).This means that their highest energy subshell is always a d-subshell. Transition metals are found in the d-block of the periodic table. The second series includes the elements yttrium (symbol Y, atomic number 39) to cadmium (symbol Cd, atomic number 48). Transition metals are elements whose atoms have a partially filled d-subshell, or which form at least one stable ion with a partially filled d-subshell of electrons.The first main transition series begins with either scandium (symbol Sc, atomic number 21) or titanium (symbol Ti, atomic number 22) and ends with zinc (symbol Zn, atomic number 30).Their properties are transitional between highly reactive metallic elements of s−block which are typically ionic compounds and elements of p−block which are large - covalent.The definition of a transition metal, and how to write the electron configuration including examples for Fe and Zn. Properties, similarities, and other facts. Transition metals are elements with an incomplete d-subshell or that can form at least one stable cation with an incomplete d-subshell. These d-block elements are called transition elements because they exhibit transitional behavior between s− and p−elements. Chemistry for Kids: Elements Kids learn about the transition metals of the periodic table. The position of d-block elements is in between s−block and p−block elements in the periodic table. The $$d$$- block elements that are the elements from the group $$3-12$$ are called transition elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
